Blood Program
Thanks to the generous donations, the Japanese Red Cross Society is able to deliver blood products to medical institutions across the country.
Blood transfusion plays an important life-saving function in the treatment of malignant neoplasms (cancer), hematological diseases and other disorders. Blood cannot, as of yet, be manufactured through artificial processes. Blood collected by way of voluntary, non-remunerated donations from healthy individuals saves the lives of countless patients on a daily basis.
To those who are considering donating blood within Japan:
Thank you very much for your support and kind consideration.
Under the blood donation policy and for safety reasons regarding the person donating blood, we are required to ask you to fill out a mandatory pre-questionnaire in Japanese, and have a certified physician conduct a preliminary medical examination for blood donation in Japanese. Both of these steps will take place on site at any of our blood donation facilities. For this reason, we cannot accept donations from individuals who are unable to understand and respond in Japanese. Thank you very much for your understanding and cooperation.
History
The JRCS is the only organization in Japan collecting and supplying blood for use in transfusions. In 1952 the Japanese Red Cross Society Tokyo Blood Bank was established and blood program operations began. This was primarily a blood deposit system that gave special consideration to donors; they would be placed on a priority list for blood transfusions in the future. The concept of non-remunerated blood donations was uncommon at the time. Instead, the practice of paid blood donations initiated by private blood banks was widespread. As a result, there were numerous occurrences of patients developing post-transfusion hepatitis after being infected by blood products collected from paid donors who had unhealthy lifestyle practices. Infected blood collected from paid donors developed into a social problem referred to as "yellow blood."
To avoid repeating the errors of the past, the JRCS, in cooperation with the national and local governments, works to maintain blood donation facilities with effective screening capabilities and strives to spread and popularize the concept of donating blood. In 1982, a transition to a genuine blood donation system based on the principle of voluntary and unpaid blood donation was carried out. Today, the blood program functions reliably and effectively through collection and distribution entities whose operations are in strict compliance with related laws (e.g. Act on Securing Quality, Efficacy and Safety of Products Including Pharmaceuticals and Medical Devices).
Major Initiatives
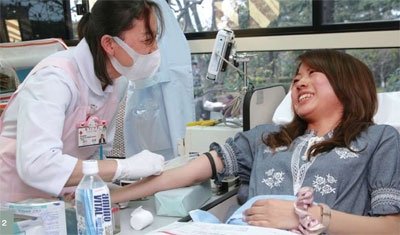
Ensuring a stable supply of blood donors is a major challenge that must be quickly addressed. In order to spread a better understanding of blood donation and thus increase the number of donors especially among the younger generation, which will form an important pillar of the future blood donation movement the JRCS works to implement various measures. These serve to secure more blood donors among the younger generation, increase the number of companies and organizations participating in group blood donations, and increase the number of people that provide multiple donations.

The JRCS is creating blood donation rooms that cater to the needs of the local community. For example, some donation centers have a child-friendly waiting room so as to encourage parents to contribute. Some centers are also being restructured to accommodate a greater number of people without jeopardizing each patient's comfort or safety. The latter is a factor that the JRCS especially takes into consideration. In order to ensure the safety of blood products, the JRCS implements a variety of safety measures. Examples of primary safety measures implemented to date include identification of the donor, retrospective studies, retaining deposits of fresh frozen plasma, improving the accuracy of nucleic acid amplification testing (NAT), removal of leukocytes, and diversion of first drawn blood prior to storage. Additionally, the JRCS is currently considering introducing techniques aimed at reducing infectious agents, as well as measures to prevent transfusion-related acute lung injury (TRALI), a serious post-transfusion adverse reaction. In 2006, a compensation system was established for blood donors who are adversely affected by the process of donating blood. Should a donor need a medical examination at a medical institution as a result of subcutaneous hemorrhaging, nerve damage, or other injuries to their well-being (secondary effects from giving blood) caused during the donation process, they are entitled to receive a fixed monetary compensation.

The JRCS will continue its efforts to improve the safety of blood products and secure a stable blood supply. In order to build an efficient and rational business plan for the future, and to carry out stable, sustainable donation programs, the JRCS initiated, in April 2013, a wide-area supply and demand management system that extends beyond prefectural boundaries while centralizing its finances.
International Cooperation
Since 1978, the JRCS has been receiving blood services trainees from sister Red Cross and Red Crescent societies mainly in the Asia region.
Many of the executives responsible for blood program in the Asian region have received this training in Japan, and it has contributed to the development of the blood program in each country and the formation of networks within the region.
We also provide technical training for countries that wish to participate in it, including training in the development of monoclonal antibody-producing cells.
In addition, a total of 8 meetings have been held between 1995 and 2017 of the Red Cross and Red Crescent Symposium on blood program in the Asia region for the purpose of contributing to the steady development of and cooperation in blood program in the region by sharing the current situation and challenges in the blood projects of each country, with a particular focus on the technical aspects among representatives. The symposia were co-sponsored by the Japanese Red Cross Society and the Thai Red Cross Society.
In view of the need for a forum where countries can exchange views candidly, the Japanese Red Cross Society and the Thai Red Cross have decided to hold a new Red Cross and Red Crescent Forum on Blood Program in Asia.





